3. 结构
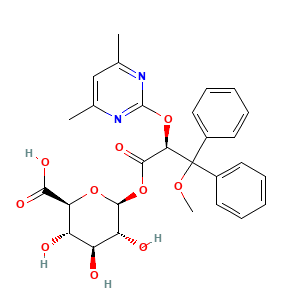
3.1 二维结构
3.2 三维结构
-1
-2
-3
70 73 0 1 0 0 0 0 0999 V2000
3.1463 -0.0472 -0.3001 O 0 0 0 0 0 0 0 0 0 0 0 0
1.1706 0.6033 0.7562 O 0 0 0 0 0 0 0 0 0 0 0 0
5.4438 1.7481 2.5931 O 0 0 0 0 0 0 0 0 0 0 0 0
2.6202 2.5130 2.2515 O 0 0 0 0 0 0 0 0 0 0 0 0
6.6077 1.0933 0.0117 O 0 0 0 0 0 0 0 0 0 0 0 0
-0.2380 -1.4302 1.7952 O 0 0 0 0 0 0 0 0 0 0 0 0
-2.0439 0.4347 -0.7636 O 0 0 0 0 0 0 0 0 0 0 0 0
6.1741 -0.7077 -1.9502 O 0 0 0 0 0 0 0 0 0 0 0 0
0.4321 1.0222 -1.3854 O 0 0 0 0 0 0 0 0 0 0 0 0
4.3083 -1.9519 -1.5418 O 0 0 0 0 0 0 0 0 0 0 0 0
-1.8006 2.7030 -0.4147 N 0 0 0 0 0 0 0 0 0 0 0 0
-3.8118 1.7859 -1.3744 N 0 0 0 0 0 0 0 0 0 0 0 0
4.7103 1.6918 1.3686 C 0 0 2 0 0 0 0 0 0 0 0 0
3.2450 1.3713 1.6654 C 0 0 2 0 0 0 0 0 0 0 0 0
5.3479 0.6195 0.4847 C 0 0 1 0 0 0 0 0 0 0 0 0
2.4867 1.0131 0.3870 C 0 0 1 0 0 0 0 0 0 0 0 0
4.4600 0.3100 -0.7142 C 0 0 1 0 0 0 0 0 0 0 0 0
-1.1557 -1.3057 0.6891 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.1219 0.2067 0.3055 C 0 0 1 0 0 0 0 0 0 0 0 0
0.2305 0.6570 -0.2340 C 0 0 0 0 0 0 0 0 0 0 0 0
4.9377 -0.9102 -1.4327 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.6563 -2.2569 -0.4145 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.5621 -1.7387 1.1318 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.3683 -2.3748 -1.6082 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.9318 -3.0821 1.0639 C 0 0 0 0 0 0 0 0 0 0 0 0
0.5084 -2.9998 -0.2209 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.4688 -0.7889 1.6025 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.8461 -1.0092 2.9997 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.9158 -3.2357 -2.6082 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.2080 -3.4755 1.4663 C 0 0 0 0 0 0 0 0 0 0 0 0
0.9608 -3.8606 -1.2209 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.7450 -1.1825 2.0050 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.5732 1.6915 -0.8543 C 0 0 0 0 0 0 0 0 0 0 0 0
0.2487 -3.9785 -2.4146 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.1146 -2.5258 1.9369 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.3539 3.9291 -0.5195 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.3033 3.0402 -1.4498 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.6133 4.1604 -1.0335 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5318 5.0783 -0.0405 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.6749 3.1894 -2.0178 C 0 0 0 0 0 0 0 0 0 0 0 0
4.8068 2.6833 0.9105 H 0 0 0 0 0 0 0 0 0 0 0 0
3.1693 0.5582 2.3971 H 0 0 0 0 0 0 0 0 0 0 0 0
5.5429 -0.2862 1.0721 H 0 0 0 0 0 0 0 0 0 0 0 0
2.4224 1.8967 -0.2636 H 0 0 0 0 0 0 0 0 0 0 0 0
4.4084 1.1335 -1.4372 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.3367 0.8707 1.1499 H 0 0 0 0 0 0 0 0 0 0 0 0
5.3591 0.8866 3.0361 H 0 0 0 0 0 0 0 0 0 0 0 0
2.6730 3.2461 1.6148 H 0 0 0 0 0 0 0 0 0 0 0 0
6.4495 1.8930 -0.5184 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.2854 -1.8212 -1.7843 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.2495 -3.8630 0.7428 H 0 0 0 0 0 0 0 0 0 0 0 0
1.0981 -2.9391 0.6885 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.2415 0.2659 1.6960 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.0418 -0.9674 3.7439 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.5501 -1.7474 3.3965 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.2637 -0.0009 2.9812 H 0 0 0 0 0 0 0 0 0 0 0 0
6.5066 -1.4990 -2.4251 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.4718 -3.3300 -3.5363 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.4933 -4.5226 1.4222 H 0 0 0 0 0 0 0 0 0 0 0 0
1.8685 -4.4381 -1.0713 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.4504 -0.4441 2.3752 H 0 0 0 0 0 0 0 0 0 0 0 0
0.6009 -4.6490 -3.1930 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.1072 -2.8325 2.2532 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.0333 5.1564 -1.1049 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.0439 5.5991 0.7748 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.5607 4.7377 0.3336 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.3500 5.7835 -0.8578 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.6504 3.8213 -2.9114 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.0904 2.2173 -2.3032 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.3460 3.6396 -1.2793 H 0 0 0 0 0 0 0 0 0 0 0 0
1 16 1 0 0 0 0
1 17 1 0 0 0 0
2 16 1 0 0 0 0
2 20 1 0 0 0 0
3 13 1 0 0 0 0
3 47 1 0 0 0 0
4 14 1 0 0 0 0
4 48 1 0 0 0 0
5 15 1 0 0 0 0
5 49 1 0 0 0 0
6 18 1 0 0 0 0
6 28 1 0 0 0 0
7 19 1 0 0 0 0
7 33 1 0 0 0 0
8 21 1 0 0 0 0
8 57 1 0 0 0 0
9 20 2 0 0 0 0
10 21 2 0 0 0 0
11 33 1 0 0 0 0
11 36 2 0 0 0 0
12 33 2 0 0 0 0
12 37 1 0 0 0 0
13 14 1 0 0 0 0
13 15 1 0 0 0 0
13 41 1 0 0 0 0
14 16 1 0 0 0 0
14 42 1 0 0 0 0
15 17 1 0 0 0 0
15 43 1 0 0 0 0
16 44 1 0 0 0 0
17 21 1 0 0 0 0
17 45 1 0 0 0 0
18 19 1 0 0 0 0
18 22 1 0 0 0 0
18 23 1 0 0 0 0
19 20 1 0 0 0 0
19 46 1 0 0 0 0
22 24 2 0 0 0 0
22 26 1 0 0 0 0
23 25 2 0 0 0 0
23 27 1 0 0 0 0
24 29 1 0 0 0 0
24 50 1 0 0 0 0
25 30 1 0 0 0 0
25 51 1 0 0 0 0
26 31 2 0 0 0 0
26 52 1 0 0 0 0
27 32 2 0 0 0 0
27 53 1 0 0 0 0
28 54 1 0 0 0 0
28 55 1 0 0 0 0
28 56 1 0 0 0 0
29 34 2 0 0 0 0
29 58 1 0 0 0 0
30 35 2 0 0 0 0
30 59 1 0 0 0 0
31 34 1 0 0 0 0
31 60 1 0 0 0 0
32 35 1 0 0 0 0
32 61 1 0 0 0 0
34 62 1 0 0 0 0
35 63 1 0 0 0 0
36 38 1 0 0 0 0
36 39 1 0 0 0 0
37 38 2 0 0 0 0
37 40 1 0 0 0 0
38 64 1 0 0 0 0
39 65 1 0 0 0 0
39 66 1 0 0 0 0
39 67 1 0 0 0 0
40 68 1 0 0 0 0
40 69 1 0 0 0 0
40 70 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
(2S,3S,4S,5R,6S)-6-[(2S)-2-(4,6-dimethylpyrimidin-2-yl)oxy-3-methoxy-3,3-diphenylpropanoyl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid
4.2 InChl
InChI=1S/C28H30N2O10/c1-15-14-16(2)30-27(29-15)39-23(25(36)40-26-21(33)19(31)20(32)22(38-26)24(34)35)28(37-3,17-10-6-4-7-11-17)18-12-8-5-9-13-18/h4-14,19-23,26,31-33H,1-3H3,(H,34,35)/t19-,20-,21+,22-,23+,26-/m0/s1
4.3 InChlKey
QBHJFBFSJYTXDX-MVTLKLODSA-N
4.4 Canonical SMILES
CC1=CC(=NC(=N1)OC(C(=O)OC2C(C(C(C(O2)C(=O)O)O)O)O)C(C3=CC=CC=C3)(C4=CC=CC=C4)OC)C
4.5 lsomeric SMILES
CC1=CC(=NC(=N1)O[C@H](C(=O)O[C@H]2[C@@H]([C@H]([C@@H]([C@H](O2)C(=O)O)O)O)O)C(C3=CC=CC=C3)(C4=CC=CC=C4)OC)C
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病